

PLN-101325: Muscular Dystrophy
Pliant is developing therapies that may directly improve muscle function and viability while reducing the reactive fibrosis that results from progressive muscle destruction.

Our Muscular Dystrophy Program
Muscular dystrophy comprises a group of inherited diseases, all characterized by inborn errors in dystrophin, a protein that is crucial to muscle function. Lack of proper dystrophin function results in damage to muscle cells, which over time leads to muscle degeneration, and replacement of muscle tissue with fat and fibrotic tissue.
Pliant has identified α7β1, a target integrin receptor that acts as a natural compensatory mechanism in Duchenne muscular dystrophy (DMD), as well as other types of muscular dystrophy. By activating this receptor, it may be possible to stabilize the muscle cells, thereby increasing muscle strength and reducing muscle damage.
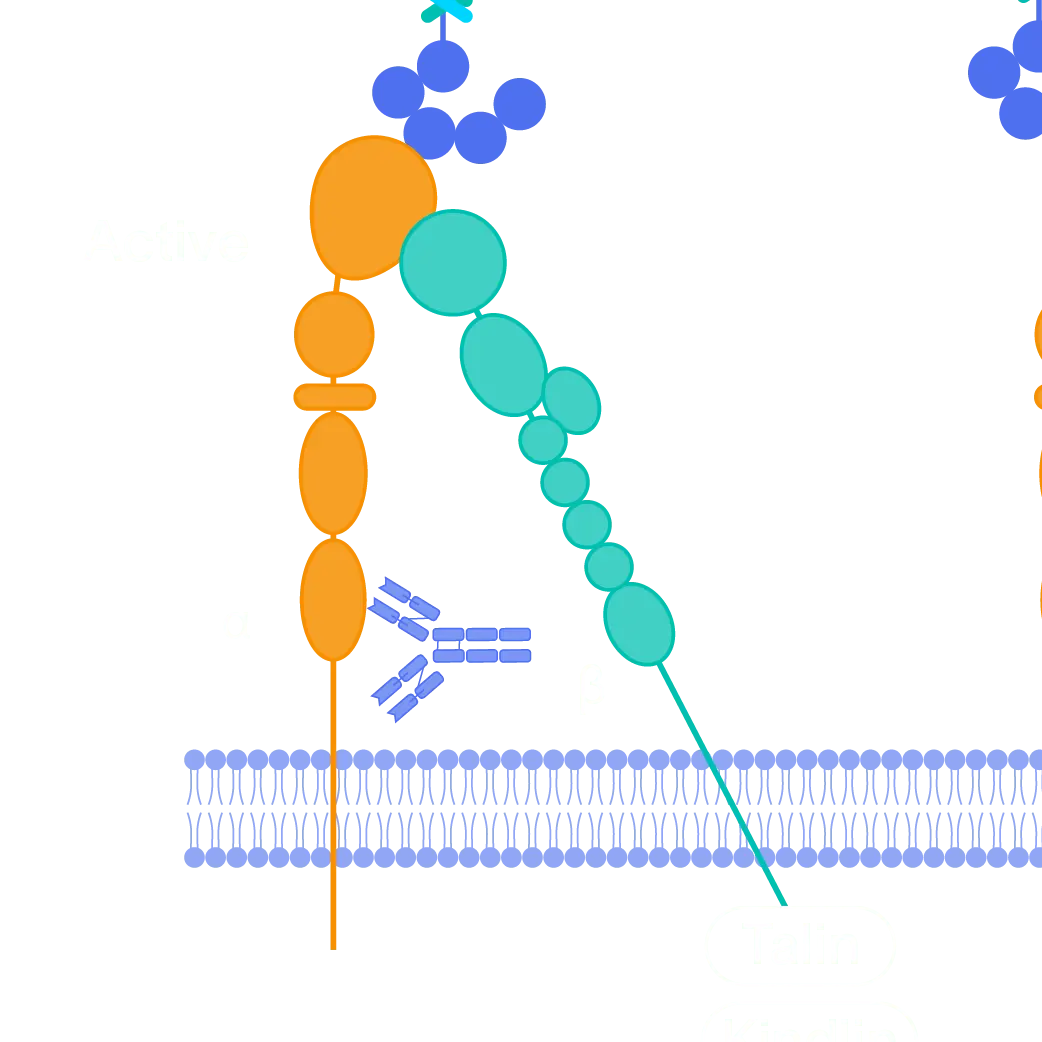
Program Highlights
- Targeting α7β1, an integrin receptor on the muscle cell surface
- α7β1 is upregulated as a compensatory mechanism in multiple types of muscular dystrophy
- α7β1 acts as a substitute for dystrophin, helping to stabilize the muscle membrane, increasing muscle strength, and decreasing muscle damage
- Mutations in the target receptor alone actually causes a human congenital myopathy similar to muscular dystrophy
- PLN-101325, an α7β1 integrin activating antibody, has the potential to combine across multiple muscular dystrophy indications because the target integrin is upregulated across multiple forms of muscular dystrophy
Development Status
- PLN-101325 is currently Phase 1 ready